Description
Process Description Water Softener- Residential
Water Softening using the Ion Exchange Process
Hard Water contains dissolved minerals in the form of Calcium (Ca), Magnesium (Mg), and Iron (Fe). Hard water has a tendency to form scale deposits in pipes, hot water systems and on plumbing fixtures. It also reduces the ability of soap to dissolve in water and will form soap scum. Removal of these minerals is accomplished by softening the water through an ion exchange process using a water softener. As the water flows through the mineral tank of the water softener, the dissolved minerals become attached to the resin, creating soft water. Over a period of time the resin in the water softener will become exhausted, and the water softener will regenerate using a brine solution produced from the salt in the brine tank.
What are the benefits of a water softener?
- Higher Quality Drinking Water
- Prevents Hard Water Scale
- Prevents Staining on Bathroom & Kitchen Fixtures as well as Dishes, Dishwasher, Washing Machine and Clothes
- Significantly Reduces Soap and Cleaning Product Consumption and eliminates soap scum.
- Provides Excellent Grooming & Cosmetic Benefits
- Reduces Water Heating Costs and extends the life of the water heater.
- Prolongs the Life of Water Heaters, Icemakers, Dishwashers, Coffeemakers and Plumbing Fixtures.
About Hardness in water
Hardness in water is caused by naturally occurring salts of calcium and magnesium which are found to varying degrees in all water sources. The higher the concentration of these salts, the “harder” the water is. Hard water gets its name from the fact that these calcium and magnesium salts react with soap and detergent forming an insoluble scum and making it “hard” to make a lather. Hard water not only makes washing with soap and detergent more difficult and costly – it also forms limescale, a rock-like deposit, when the water is heated. This limescale reduces heat transfer and makes heat exchange equipment more costly to operate, and in extreme cases such as a steam boiler can lead to catastrophic failure.
.
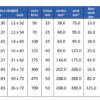
| Water Hardness Scale |
| Grains Per Gallon |
Milligrams Per Liter (mg/L)or Parts Per Million (ppm) |
Classification |
| less than 1.0 |
less than 17.1 |
Soft |
| 1.0 – 3.5 |
17.1 – 60 |
Slightly Hard |
| 3.5 – 7.0 |
60 – 120 |
Moderately Hard |
| 7.0 – 10.5 |
120 – 180 |
Hard |
| over 10.5 |
over 180 |
Very Hard |
What problems does limescale cause?
Increased Energy Costs – The scale acts as insulation, dramatically reducing heating or cooling efficiency and adding to your energy costs. In many industrial and commercial applications a cooling water softener or boiler feed water softener will rapidly pay for itself in reduced energy costs.
Increased Pumping Costs and Wear – Scale reduces the internal bore of pipework, which causes impaired flow and increased pumping costs as the pumps must work harder to deliver the required water volume.
Overheating and Hot-Spots – Because scale is an insulator, it can cause hot spots to form on heat transfer surfaces as heat is not conducted away fast enough. In a steam boiler this over-heating can cause potentially disastrous failure, making an efficient boiler water softener an essential part of the feed water treatment system.
Our range of water softeners with Clack Control Valves.
Our water softeners come with world renowned Clack Valves for reliable operation. Our water softeners and our Clack Valves are made in the USA, which have a huge advantage over cheaper Chinese made products. Our water softeners superiority has been proven over years of application. The Clack Softener Valve features a versatile multi-function control that enables programming to either Timer or Water-Meter Demand Regeneration. Demand Regeneration is the most efficient and popular softening method. An in-built water meter monitors the actual water flow and usage patterns and initiates a regeneration only when required. This offers significant salt and water savings over timer regeneration softeners. The control includes a Coin Cell Lithium battery back-up with an 8 hour carry over to save day and time settings in event of blackout. The control uses safe 12 volt power from the wall-mount transformer supplied.
Advantages of our water softeners:
- Will regenerate based on actual water treated and taking into account water hardness.
- Will regenerate at a pre-set time interval when water usage is low.
- Salt usage is optimised based on using settings taking into account actual water hardness and flow rate.
- We use only premium water softening resin that is resistant to chlorine degradation.
Click link below for our water softener range:
Water Softener Range